What will be the final temperature attained if all the heat released in neutralization of 1L of 0.2M NH4OH with 2L of 0.1M HCl increases the temperature of the final solution having density 0.95 g/mL and specific heat capacity = J/g°C if original temperature was 27°C. Assume weak base to be completely unionized.
Know your College Admission Chances Based on your Rank/Percentile, Category and Home State.
Get your JEE Main Personalised Report with Top Predicted Colleges in JoSA
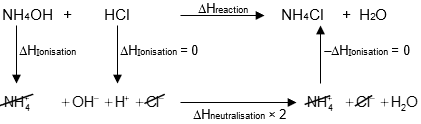
ΔHreaction = ΔHionisatuionWB + ΔHneutralisation
ΔHreaction = (+10 – 57.5) kJ for unit extent of reaction = –47.5 kJ
mol of NH4OH = 0.2
mol of HCl = 0.2 No limiting reagent
Released heat = 0.2 × 47.5 kJ = 9.5 kJ QP = msdT
9.5 ×1000 J = 3000 mL × 0.95 g/mL × J/g .K × dT
dT = 10
T2 = 310 K