Engineering
Chemistry
Colloids Preparation and Properties
Question
Which of the following are correct statements?
Know your College Admission Chances Based on your Rank/Percentile, Category and Home State.
Get your JEE Main Personalised Report with Top Predicted Colleges in JoSA
Solution
(A) During ester hydrolysis an acid is formed as product so (H+) increases and rate also increases.
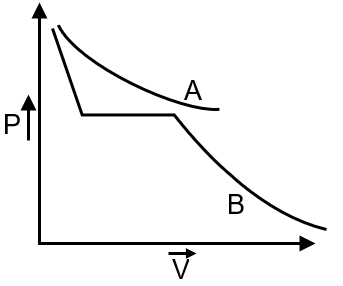
(B) From graph critical temperature of B > A so extent of adsorption of B will be more
(C) Correct because in colloids dispersed phase and dispersion medium have opposite chargers.
(D) As fraction of oil increases viscosity increases.