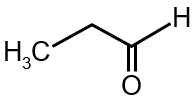
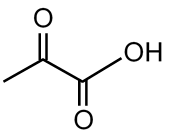
Which of the following compound can not show haloform reaction.
Know your College Admission Chances Based on your Rank/Percentile, Category and Home State.
Get your JEE Main Personalised Report with Top Predicted Colleges in JoSA
The haloform reaction occurs with methyl ketones (R-CO-CH3) or compounds that can be oxidized to methyl ketones, like ethanol and acetaldehyde. The key structural requirement is a group attached to a carbonyl carbon or an alcohol that can form it. The compound that cannot show this reaction is the one lacking this methyl group. In the given options, the third structure (cyclopentanone) does not have a methyl group attached to its carbonyl; it is a cyclic ketone without the -CO- moiety.
Final Answer: The third compound (cyclopentanone) cannot show the haloform reaction.