Engineering
Chemistry
Hydrogen Bonding
Structure of Some Important Compounds
Vapour Pressure
Question
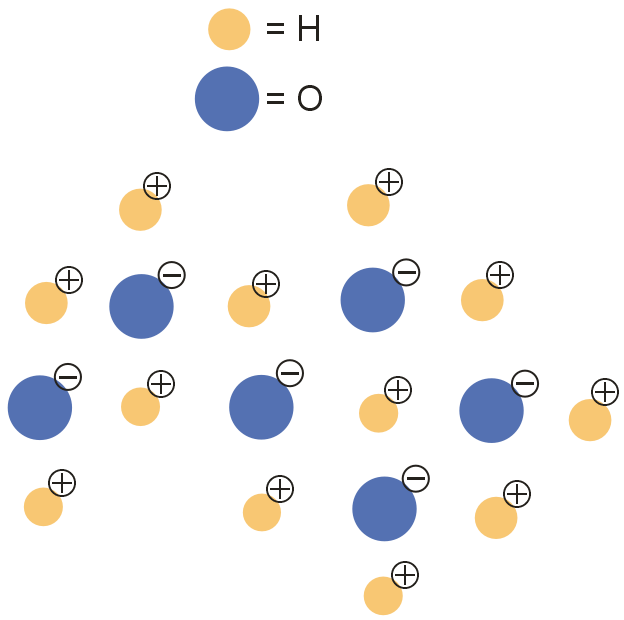
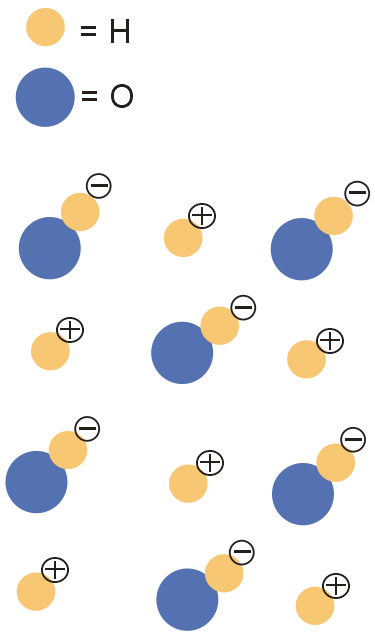
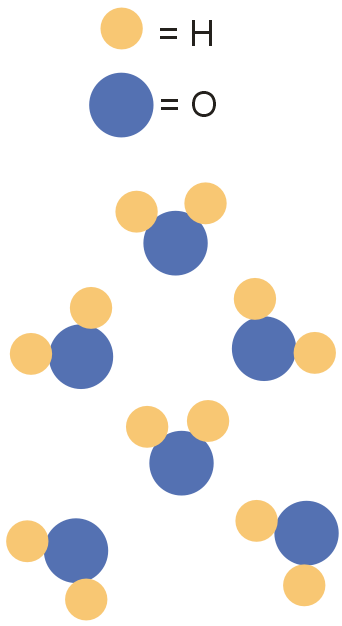
Which of the following particle diagrams BEST represents the structure of ice, H2O(s)?
Know your College Admission Chances Based on your Rank/Percentile, Category and Home State.
Get your JEE Main Personalised Report with Top Predicted Colleges in JoSA
Solution
The atomic structure of a water (or dihydrogen monoxide) molecule consists of two hydrogens (H) atoms joined to one oxygen (O) atom. The unique way in which the hydrogen atoms are attached to the oxygen atom causes one side of the molecule to have a negative charge and the area in the opposite direction to have a positive charge. The resulting polarity of charge causes molecules of water to be attracted to each other forming strong molecular bonds.