Engineering
Chemistry
Isomerism in Coordination Compounds
Geometrical Isomerism
Optical Isomerism
Question
Which of the following statement is true?
Know your College Admission Chances Based on your Rank/Percentile, Category and Home State.
Get your JEE Main Personalised Report with Top Predicted Colleges in JoSA
Solution
(A) Both cis and trans isomers of [PtCI2(NH3)2]2+ contain a plane of symmetry and are optically inactive.Thus statement A is incorrect.
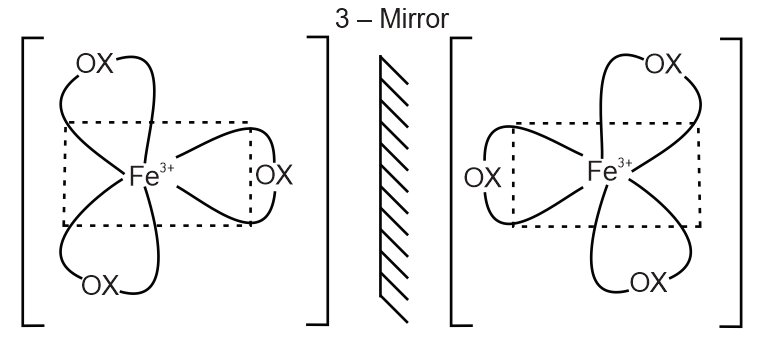
(B) The complex [Fe(C2O4)3]3– is of the type [M(AA)3]n±.It contains no symmetry element and exhibits only optical isomerism.Geometrical isomerism is not possibleThus statement B is correct.
(C) In square planar [Mabcd] complex,plane of symmetry is present. Hence,they do not show optical isomerism. In this complex three geometrical isomers are possible. Thus statement C is incorrect.
(D) In [Mabcd] tetrahedral complex, no element of symmetry is present.
(B) The complex [Fe(C2O4)3]3– is of the type [M(AA)3]n±.It contains no symmetry element and exhibits only optical isomerism.Geometrical isomerism is not possibleThus statement B is correct.
(C) In square planar [Mabcd] complex,plane of symmetry is present. Hence,they do not show optical isomerism. In this complex three geometrical isomers are possible. Thus statement C is incorrect.
(D) In [Mabcd] tetrahedral complex, no element of symmetry is present.
Hence,optical isomerism is possible.Hence,the statement D is incorrect.